Mar 22 2022
NLS Pharmaceutics Announces Positive Interim Top-Line Data for Quilience® (Mazindol ER) in Patients with Narcolepsy
h in News

– Clinically meaningful results with Mazindol ER exhibiting a 39% reduction in ESS from baseline (-4.3 points placebo-adjusted); confirms statistical powering of the ongoing Phase 2a trial
– Safety and tolerability data consistent with prior mazindol clinical trials, no reported SAEs
– 82% of patients that completed the randomized trial to date have rolled over into the open label extension (OLE) study
– Majority of patients that enrolled in the OLE (both NT1 and NT2) remain in the study and on Mazindol ER monotherapy
Switzerland/Zurich, March 16, 2022 – NLS Pharmaceutics Ltd. (Nasdaq: NLSP, NLSPW) (“NLS” or the “Company”), a Swiss clinical-stage biopharmaceutical company focused on the discovery and development of innovative therapies for patients with rare and complex central nervous system disorders, announces positive interim top-line data from its Phase 2a clinical trial evaluating its lead product candidate, Quilience® (Mazindol ER), in the treatment of narcolepsy. The data are being presented and discussed today during a symposium sponsored by NLS at the World Sleep Congress (World Sleep 2022), being held in Rome, Italy.
Quilience (Mazindol ER) Interim Efficacy Data Summary
Of the 60 patients targeted for full enrollment in the U.S. Phase 2a trial (Study NLS-1021), 27 were randomized and completed the study for the interim analysis. The interim analysis database included 13 patients on treatment and 14 patients on placebo, with balanced mean baseline Epworth Sleepiness Scale (ESS) scores (18.6 +/-2.81 for treatment and 18.0 +/-2.72 for placebo). All patients enrolled in the interim analysis were utilizing combination therapy before the trial’s wash out period, with the majority of participants on stimulant or wake-promoting agents to treat their excessive daytime sleepiness (EDS). Approximately one-third of patients enrolled in the interim analysis were diagnosed with narcolepsy type 1 (NT1) and suffer from both EDS and cataplexy symptoms. Eligible NT1 patients must have moderate to severe disease according to the study protocol – defined as having more than 3-4 cataplexy attacks per week. Study participants on treatment are required to undergo a 1-2-week wash out period (depending on prior therapy). After the wash out period, participants are randomized to receive either once-daily treatment with Mazindol ER 2mg for week 1 and 3mg for weeks 2-4, or placebo for 4 weeks.
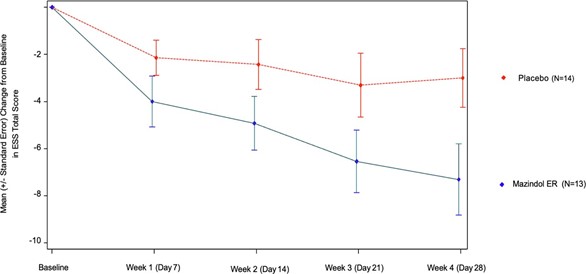
For EDS, the trial’s primary endpoint, patients on Mazindol ER exhibited a mean decrease of 7.3 points on the ESS compared to a 3.0 point decrease for patients on placebo. Patients on Mazindol ER showed a mean 39% improvement on ESS from baseline. The placebo-adjusted reduction of 4.3 points is deemed to be clinically meaningful and is competitive compared to current narcolepsy treatments, despite the short duration and relatively small number of patients in the interim analysis. These interim results confirm the statistical power of the ongoing Phase 2a trial, which will continue as planned.
In the figure below, a rapid onset of action is observed for Mazindol ER, with treated patients exhibiting consistent and sustained EDS improvement over the 4-week treatment period. The placebo
effect in the interim analysis exhibits stabilization after week 3, consistent with other narcolepsy therapeutic clinical trials.1,2.
Epworth Sleepiness Scale (ESS) Total Score: Change from Baseline to Last Visit (ITT Population)
Quilience (Mazindol ER) Phase 2a Interim Safety/Tolerability Data Summary
To date, Mazindol ER was generally safe and well-tolerated in the current Phase 2a trial, and the interim data are consistent with prior mazindol studies, including the Company’s trials evaluating treatment of attention deficit hyperactive disorder (ADHD). No patients dropped out of the study due to safety or efficacy concerns, and only one patient in the interim analysis discontinued the study for personal reasons. This compares favorably to other clinical trials for narcolepsy treatments, for which study discontinuation rates can be as high as 15% or more for efficacy and safety reasons.3 No serious adverse events were reported in the interim analysis and all reported adverse events were mild or moderate and resolved immediately with no intervention required.
“These interim data are very encouraging and demonstrate the potential for Mazindol ER to address the core symptoms of narcolepsy with a rapid onset of action and a favorable safety and tolerability profile,” said Bruce Corser, M.D., Medical Director at the Cincinnati-based Sleep Management Institute and a clinical investigator in the NLS-1021 trial. “Mazindol’s dual mechanism of action as a partial orexin agonist and pan-monoaminergic reuptake inhibitor distinguishes this therapeutic candidate and is of particular interest in the field given the drug’s potential to treat narcolepsy at its root cause. I am looking forward to the final results from this trial and the potential to utilize this novel therapy in more of my patients.”
Quilience (Mazindol ER) Open Label Extension (OLE) Study Update
Study NLS-1022 (OLE study) enables patients completing the randomized controlled trial to access treatment with Mazindol ER without any background stimulant and or anti-cataplexy treatment for up to 6 months. As of the data collection date for the Phase 2a interim analysis (February 25, 2022), 82% of patients who completed the randomized controlled study elected to participate in the OLE study. A majority of those patients remain in the OLE study and on treatment with Mazindol ER, including participants suffering from NT1, with one patient on single agent therapy for over 3 months.
“We are extremely pleased with these interim results, which offer the first evidence from a prospective randomized clinical trial that Mazindol ER has the potential to become a disruptive treatment for narcolepsy,” said Alex Zwyer, Chief Executive Officer of NLS Pharmaceutics. “I am very proud of the NLS clinical team’s execution and thankful for our clinical sites who are recruiting participants at among the fastest pace in the industry as we focus on bringing this important treatment to patients as soon as possible. Importantly, we are seeing strong participation from NT1 patients in this trial. Given that the interim results are in-line with the documented historical utility of mazindol in treatment-resistant narcolepsy patients under compassionate use, we are confident that our proprietary Mazindol ER formulation will prove to be a safe and effective therapy that can address a broad spectrum of narcolepsy symptoms.”
“These interim results have exceeded on our expectations with regard to the beneficial effects of Mazindol ER’s unique mechanism of action and validate the body of retrospective evidence that support the compound’s efficacy and safety in the treatment of narcolepsy,” said Eric Konofal, PhD, MD Chief Scientific Officer of NLS Pharmaceutics. “The rapid onset of action and large effect size between treatment and placebo are superb in this dataset and are consistent with the results that we generated in our two ADHD trials, in addition to affirming Mazindol ER’s benign safety and tolerability profile. Mazindol is thought to address a wide spectrum of narcolepsy symptoms including EDS, cataplexy, sleep paralysis, and nocturnal hallucinations, and we are excited to obtain the final Phase 2a results to fully understand the drug’s therapeutic potential, and to move this product candidate into advanced clinical trials.”
About World Sleep 2022
The international Congress of the World Sleep Society is the most important meeting of Sleep Research Scientists worldwide. Now in its 16th iteration, the World Sleep Congress consistently gathers the best minds in sleep medicine and research for multiple days of scientific sessions and networking. World Sleep 2022 will be held in Rome, Italy, March 11–16, 2022, with fourteen keynote speakers selected to represent a broad range of sleep medicine and research. Twenty-two sleep medicine and research courses are in development, and attendees will have access to presentations by world-class researchers in chrono-medicine, narcolepsy, sleep surgery, insomnia, sleep-focused basic research, and more. A truly global meeting, 77 countries were represented at World Sleep 2019 which featured 89 symposia, 192 oral abstracts, and over 1,100 poster presentations. World Sleep 2022 presents a unique opportunity for sleep medicine professionals no matter their specialty or career stage.
About NLS Pharmaceutics Ltd.
NLS Pharmaceutics Ltd. is a Swiss clinical-stage biopharmaceutical company led by an experienced management team with a track record of developing and repurposing product candidates to treat rare and complex central nervous system disorders. The Company’s lead product candidate, Quilience, is a proprietary extended-release formulation of mazindol (mazindol ER) and is being developed for the treatment of narcolepsy, and potentially other sleep-wake disorders such as idiopathic hypersomnia. Mazindol is a triple monoamine reuptake inhibitor and partial orexin-2 receptor agonist, which was used for many years to treat patients diagnosed with narcolepsy in compassionate use programs. A Phase 2a clinical trial evaluating Quilience in adult subjects suffering from narcolepsy is currently ongoing in the United States. Previously, NLS successfully completed a phase 2 study in the U.S. evaluating Nolazol® (mazindol controlled-release) in adult subjects suffering from ADHD. The study met all primary and secondary endpoints and Nolazol® was well-tolerated. Quilience has received Orphan Drug Designations both in the U.S. and in Europe for the treatment of narcolepsy. Up to 1/3 of narcoleptic patients are also diagnosed with ADHD.
Safe Harbor Statement
This press release contains express or implied forward-looking statements pursuant to U.S. Federal securities laws. For example, NLS is using forward-looking statements when it discusses that the interim results confirm the power of the ongoing Phase 2a trial and demonstrate the potential for Mazindol ER, that the trial will continue as planned, that Mazindol ER has the potential to be used in more patients and be a disruptive treatment for narcolepsy, the belief that the interim clinical trial results confirm the beneficial effects of Mazindol ER’s unique mechanism of action and validate the body of retrospective evidence that support the compound’s efficacy and safety in the treatment of narcolepsy, the confidence that its Mazindol ER formulation will prove to be a safe and effective therapy that can address a broad spectrum of narcolepsy symptoms and the timing of the final results of the trial. These forward-looking statements and their implications are based on the current expectations of the management of NLS only, and are subject to a number of factors and uncertainties that could cause actual results to differ materially from those described in the forward-looking statements. The following factors, among others, could cause actual results to differ materially from those described in the forward-looking statements: changes in technology and market requirements; NLS may encounter delays or obstacles in launching and/or successfully completing its clinical trials; NLS’ products may not be approved by regulatory agencies, NLS’ technology may not be validated as it progresses further and its methods may not be accepted by the scientific community; NLS may be unable to retain or attract key employees whose knowledge is essential to the development of its products; unforeseen scientific difficulties may develop with NLS’ process; NLS’ products may wind up being more expensive than it anticipates; results in the laboratory may not translate to equally good results in real clinical settings; results of preclinical studies may not correlate with the results of human clinical trials; NLS’ patents may not be sufficient; NLS’ products may harm recipients; changes in legislation may adversely impact NLS; inability to timely develop and introduce new technologies, products and applications; loss of market share and pressure on pricing resulting from competition, which could cause the actual results or performance of NLS to differ materially from those contemplated in such forward-looking statements. Except as otherwise required by law, NLS undertakes no obligation to publicly release any revisions to these forward-looking statements to reflect events or circumstances after the date hereof or to reflect the occurrence of unanticipated events. More detailed information about the risks and uncertainties affecting NLS is contained under the heading “Risk Factors” in NLS’ annual report on Form 20-F for the year ended December 31, 2020 filed with the Securities and Exchange Commission (SEC), which is available on the SEC’s website, www.sec.gov, and in subsequent filings made by NLS with the SEC.
References:
[1] Kollb-Sielecka et al. The European Medicines Agency Review of Pitolisant for Treatment of Narcolepsy: Summary of the Scientific Assessment by the Committee for Medicinal Products for Human Use. Sleep Medicine February 2017;(33):125-129
[2] Black J; Houghton WC; and the Xyrem® International Study Group. Sodium Oxybate Improves Excessive Daytime Sleepiness in Narcolepsy. SLEEP 2006;29(7):939-946
[3] Thorpy, M et al. A Randomized Study of Solriamfetol for Excessive Sleepiness in Narcolepsy. ANN NEUROL 2019;(85):359–370
Corporate Contact:
Alex Zwyer, CEO: +41 44 512 21 50
Investor Relations Contact:
Cindy Rizzo: +1 908-229-7050
Media Contact: Alpha Bronze, LLC:
Pascal Nigen: +1 917-385-2160
##
